- HOME
- Enzyme List
- AGH-211 α-GLUCOSIDASE(MALTASE)
-
AGH-211
α-GLUCOSIDASE(MALTASE) from Microorganism
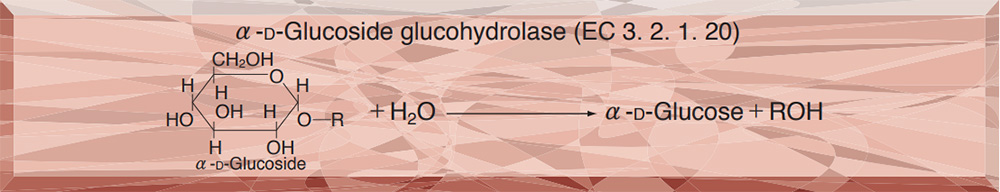
PREPARATION and SPECIFICATION
| Appearance | White amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅡ 20 U/mg-solid or more | |
| Contaminants | α-amylase | ≤1.0×10-5 % |
| Stabilizers | BSA | |
PROPERTIES
| Stability | Stable at −20 ℃ for at least one year(Fig.1) | ||
|---|---|---|---|
| Molecular weight | approx. 65,000 (Gel-filtration and SDS-PAGE) | ||
| Isoelectric point | 5.2 | ||
| Michaelis constant | 6.3×10-4M (p-Nitrophenyl-α-D-glucopyranoside) | ||
| Inhibitors | Ag+, Hg2+, PCMB, MIA | ||
| Optimum pH | 6.0−7.0(Fig.4) | ||
| Optimum temperature | 60 ℃(Fig.5) | ||
| pH Stability | pH 5.0−9.0(Fig.6) | ||
| Effect of various chemicals | (Table 1) | ||
| Thermal stability | below 60℃ (pH 7.0, 15min)(Fig.7) | ||
| Substrate* | Relative hydrolysis rate** | Substrate* | Relative hydrolysis rate** |
|---|---|---|---|
| PNPG | 100.0 | Maltose | 271.0 |
| PNPG2 | 205.0 | Maltotriose | 203.0 |
| PNPG3 | 284.0 | Maltotetraose | 168.0 |
| PNPG5 | 164.0 | Maltopentaose | 100.0 |
* : Substrate concn. 2.2mM
** : Glucose-forming activity, pH 6.8 at 37℃
APPLICATIONS
This enzyme is useful for structural investigation of carbohydrates and for enzymatic determination of α-amylase when in combination hexokinase (HXK-311) and G-6-P dehydrogenase (G6D-311, G6D321) in clinical analysis.
ASSAY
Principle
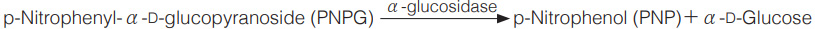
The formation of p-nitrophenol is measured at 400 nm by spectrophotometry.
Unit definition
One unit causes the formation of one micromole of PNP per minute under the conditions detailed below.
Method
Reagents
| A. 0.1M Phosphate buffer, pH 7.0 (at 25℃) | |
|---|---|
| B. PNPG solution | 20 mM (603mg P-Nitrophenyl-α-D-glucopyranoside /100 mL of H2O)(Stable for two weeks if stored at 0−5℃) |
| C. Na2CO3 solution | 0.2 M (21.2g Na2CO3 /1,000 mL of H2O) |
| D. Enzyme diluent | 0.2 M K-phosphate buffer, pH 7.0 containing 1mM of EDTA and 0.05 % of Tween 20 |
Procedure
1. Prepare the following reaction mixture in a test tube and equilibrate at 37℃ for approximately 5 minutes.
| 1.0 mL | 0.1 M phosphate buffer | (A) |
| 0.5 mL | Substrate solution | (B) |
| Concentration in assay mixture | |
|---|---|
| Phosphate buffer | 0.1 M |
| PNPG | 5.0 mM |
| EDTA | 0.25 mM |
| Tween 20 | 0.125 mg/mL |
2. Add 0.5 mL of the enzyme solution* and mix.
3. After exactly 15 minutes at 37℃, add 2.0 mL of Na2CO3 solution (C) to stop the reaction and measure the optical density at 400nm against water (OD test).
At the same time, prepare the blank by mixing the reaction mixture with 2.0 mL of Na2CO3 solution (C) after incubation for 15 minutes at 37℃, followed by the addition of the enzyme solution (OD blank).
*Dissolve the enzyme preparation in ice-cold enzyme diluent (D) and dilute to 0.006−0.022U/mL with the same buffer, immediately before the assay.
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/mL) =
-
ΔOD (OD test−OD blank)×Vt×df
18.1×t×1.0×Vs
= ΔOD×0.0295×df
Weight activity (U/mg) = (U/mL)×1/C
| Vt | : Total volume (4.0 mL) |
| Vs | : Sample volume (0.5 mL) |
| 18.1 | : Millimolar extinction coefficient of p-nitrophenol under the assay condition (cm2/micromole) |
| 1.0 | : Light path length (cm) |
| t | : Reaction time (15 minutes) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/mL) |
REFERENCES
1) Y.Suzuki, M.Shinji and N.Eto; Biochim.Biophys.Acta., 787, 281 (1984).
2) Y.Takii, K.Daimon and Y.Suzuki; Appl.Microbiol.Biotechnol., 38, 243 (1992).
3) Y.Takii, K.Takahashi, K.Yamamoto, Y.Sogabe and Y.Suzuki; Appl.Microbiol.Biotechnol., 44, 629 (1996).
Table 1. Effect of Various Chemicals on α-Glucosidase
[The enzyme dissolved in 10mM phosphate buffer, pH 7.0 contg. 0.2 % of BSA (5 U/mL) was incubated with each chemical at 25 ℃ for 1 hr.]
-
Chemical Concn.(mM) Residual
activity(%)None - 100 Metal salt 2.0 MgSO4 97 CaCl2 71 Ba(OAc)2 106 FeCl2 50 CoCl2 63 MnCl2 69 ZnCl2 104 CdCl2 47 NiCl2 110 CuSO4 39 Pb(OAc)2 75 AgNO2 0.3 HgCl2 1.2 2-Mercaptoethanol 2.0 111 PCMB 1.0 1.3 -
Chemical Concn.(mM) Residual
activity(%)MIA 2.0 0.8 NEM 2.0 120 IAA 2.0 106 Hydroxylamine 2.0 115 EDTA 5.0 112 o-Phenanthroline 2.0 114 α,α′-Dipyridyl 1.0 122 Borate 50 119 NaF 2.0 118 NaN3 2.0 123 Triton X-100 0.10 % 123 Brij 35 0.10 % 121 Tween 20 0.10 % 124 Span 20 0.10 % 43 Na-cholate 0.10 % 102 SDS 0.05 % 10 DAC 0.05 % 124
Ac, CH3CO; PCMB, p-Chloromercuribenzoate; MIA, Monoiodoacetate; NEM, N-Ethylmaleimide; IAA, Iodoacetamide; EDTA, Ethylenediaminetetraacetate; SDS, Sodium dodecyl sulfate; DAC, Dimethylbenzylalkylammonium chloride.
-
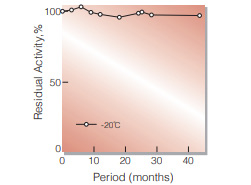
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
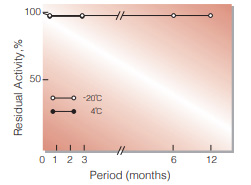
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
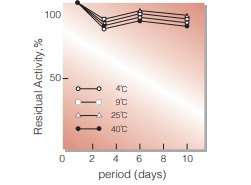
Fig.3. Stability (Liquid form)
in 50mM PIPES buffer solution, pH7.0 (contg. 0.5mM CaCl2, 0.1 % detergent) enzyme concn,:5U/mL
-
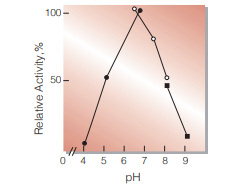
Fig.4. pH-Activity
37℃, 15 min-reaction in 100mM buffer solution: ●, pH4.0-6.0 acetate ; O, pH6.0-8.0, phosphate; ■, pH8.0-9.0, borate
-
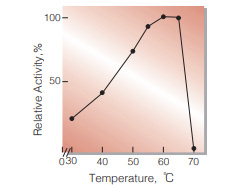
Fig.5. Thermal activity
15 min-reaction in 100mM phosphate buffer, pH7.0
-
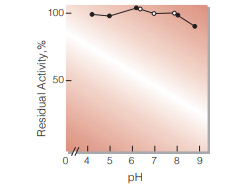
Fig.6. pH-Stability
25℃, 20hr-treatment with 50mM buffer solution contg; 0.2 % of BSA: ●, pH4.0-6.0 acetate: ○, pH6.0-8.0, phosphate; ■, pH8.0-9.0, borate. enzyme concn. : 5U/mL
-
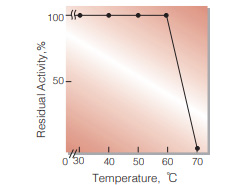
Fig.7. Thermal stability
15min-treatment with 0.2M K-phosphate buffer, pH7.0 contg. 1mM EDTA and 0.05 % Tween20. enzyme concn.: 5U/mL
活性測定法(Japanese)
1. 原理
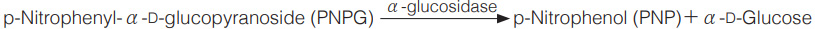
p-Nitrophenolの生成量を400nmの吸光度変化で測定する。
2.定義
下記条件下で1分間に1マイクロモルのp-Nitrophenolを生成する酵素量を1単位(U)とする。
3.試薬
- 0.1M リン酸緩衝液, pH 7.0 (25℃)
- 20mM PNPG水溶液〔603mgのP-ニトロフェニル-α-D-グルコピラノシドを100㎖の蒸留水に攪拌溶 解する〕(1~5℃保存で2週間は使用可能)
- 0.2M Na2CO3溶液(21.2gの無水炭酸ナトリウムを蒸留水に溶解し1,000 mLとする)
酵素溶液:酵素標品を予め氷冷した1mM EDTA・2Naと0.05 %Tween20を含む0.2Mリン酸緩衝液,pH7.0で溶解し0.006〜0.022U/mLに希釈する。
4.手順
1.試験管に下記反応混液を調製し,37℃で約5分間予備加温する。
| 1.0 mL | リン酸緩衝液, pH 7.0 | (A) |
| 0.5 mL | 基質溶液 | (B) |
2.酵素溶液を0.5 mLを加え,反応を開始する。
3.37℃で正確に15分間反応させた後,Na2CO3溶液(C)2.0 mL加えて反応を停止させる。この液につき400nmにおける吸光度を測定する(OD test)。
4.盲検は反応混液①を37℃で15分間放置後,Na2CO3溶液(C) 2.0 mLを加えて混和し,次いで酵素溶液0.5 mLを加えて調整する。以下同様に吸光度を測定する(ODblank)。
5.計算式
U/mL =
-
ΔOD (OD test−OD blank)×4.0(mL)×希釈倍率
18.1×1.0×15(分)×0.5(mL)
| = ΔOD×0.0295×希釈倍率 | |
| U/mg | = U/mL×1/C |
| 18.1 | : p-Nitrophenolの上記測定条件下でのミリモル分子吸光係数(cm2/micromole) |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/mL) |
CONTACT
-
For inquiries and cosultations regarding our products, please contact us through this number.
- HEAD OFFICE+81-6-6348-3843
- Inquiry / Opinion
