- HOME
- Enzyme List
- CHO-301 CHOLINE OXIDASE
CHO-301
CHOLINE OXIDASE from Microorganism

PREPARATION and SPECIFICATION
| Appearance | Yellowish amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅢ 10 U/mg-solid or more (containing approx. 20 % of stabilizers) |
|
| Contaminant | Catalase | ≤1.0×102 % |
| Stabilizers | EDTA, BSA, amino acids (glycine, sodium glutamate, etc.) | |
PROPERTIES
| Stability | Stable at −20 ℃ for at least one year (Fig.1) |
|---|---|
| Molecular weight | approx. 95,000 |
| Isoelectric point | 4.1±0.1 |
| Michaelis constants | 2.84×10-3 M (Choline), 5.33×10-3 M (Betaine aldehyde) |
| Structure | One mol of FAD is covalently bound to mol of the enzyme 8) |
| Inhibitors | p-Chloromercuribenzoate, Cu2+,Co2+,Hg2+,Ag+ |
| Optimum pH | 8.0−8.5(Fig.4) |
| Optimum temperature | 40−45 ℃(Fig.5) |
| pH Stability | pH 7.0−9.0 (30 ℃, 2 hr)(Fig.6) |
| Thermal stability | below 37℃ (pH 7.5, 10 min)(Fig.7) |
| Effect of various chemicals | (Table 1) |
APPLICATIONS
This enzyme is useful for enzymatic determination of phospholipids in combination with phospholipase D and for measuring choline esterase activity in clinical analysis.9〜11)
ASSAY
Principle

The formation of quinoneimine dye is measured at 500 nm by spectrophotometry.
Unit definition
One unit causes the formation of one micromole of hydrogen peroxide (half a micromole of quinoneimine dye) per minute under the conditions detailed below.
Method
Reagents
| A. Choline chloride solution | 2.1 %[2.1g of choline chloride/100 mL of Tris-HCl buffer (D)](Should be prepared fresh) |
|---|---|
| B. 4-AA solution | 1.0 % (1.0 g of 4-aminoantipyrine/100 mL of H2O)(Store at 4 ℃ in a brownish bottle) |
| C. Phenol solution | 1.0 % (1.0 g of phenol/100 mL of H2O; store at 4 ℃ in a brownish bottle) |
| D. Tris-HCl buffer | 0.1 M Tris-HCl buffer, pH 8.0[Dissolve 12.1 g of Tris (MW=121.14) in approx. 800 mL of H2O and, after adjusting the pH to 8.0 at 25℃ with 2.0 N HCl, make up to 1,000 mL with H2O.] |
| E. Enzyme diluent | 10 mM Tris-HCl buffer, pH 8.0, containing 2mM EDTA and 1.0 % KCl. |
Procedure
1.Prepare the following working solution (100 mL) in a brownish bottle before use and store on ice.
| 97 mL | Substrate solution | (A) |
|---|---|---|
| 1.0 mL | 4-AA solution | (B) |
| 2.0 mL | Phenol solution | (C) |
| 5.0 mL | Peroxidase from horseradish (110 purpurogallin units/mg) (Toyobo GradeⅢ) |
|
| Concentration in assay mixture | |
|---|---|
| Tris buffer | 97 mM |
| Choline chloride | 0.14 M |
| EDTA | 33 μM |
| KCI | 2.2 mM |
| 4-Aminoantipyrine | 0.48 mM |
| Phenol | 2.1 mM |
| POD | approx. 4.92 U/mL |
2.Pipette 3.0 mL of working solution into a cuvette (d = 1.0 cm) and equilibrate at 37 ℃ for approx. 5 minutes.
3.Add 0.05 mL of the enzyme solution* and mix by gentle inversion.
4.Record the increase in optical density at 500 nm against the working solution for 3 to 4 minutes in a spectrophotometer thermostated at 37 ℃, and calculate the ΔOD per minute from the initial linear portion of the curve.
*Dissolve the enzyme preparation in ice-cold Tris-HCl buffer (D) and dilute to 0.1−0.5 U/mL with enzyme diluent (E).
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/mL) =
-
ΔOD/min×Vt×df
12.0×1/2×1.0×Vs
= ΔOD/min×10.17×df
Weight activity (U/mg) = (U/mL)×1/C
| Vt | : Total volume (3.05 mL) |
| Vs | : Sample volume (0.05 mL) |
| 12.0 | : Millimolar extinction coefficient of quinoneimine dye under the assay conditions(cm2/micromole) |
| 1/2 | : Factor based on the fact that one mole of H2O2 produces half a mole of quinoneimine dye. |
| 1.0 | : Light path length (cm) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/mL) |
REFERENCES
1)P.J.G Mann and J.H.Quastel; Biochem.J.,31, 869 (1937).
2)P.J.G Mann et al; Biochem.J.,32, 1024 (1938).
3)H.S.Shieh; Can.J.Microbiol., 10, 837 (1964).
4)H.S.Shieh; Ibid.,11, 375 (1965).
5)G.J.J.Kortstee; Arch.Mikirobiol., 71, 235 (1970).
6)S.Ikuta, K.Matuura, S.Imamura, H.Misaki, Y.Horiuti; J.Biochem., 82, 157 (1977).
7)S.Ikuta, S.Imamura, H.Misaki and Y.Horiuti ; J.Biochem., 82, 1741 (1977).
8)M.Ohta-Fukuyama, Y.Miyake, S.Emi and T.Yamano; J.Biochem., 88, 197 (1980).
9)M.Takayama et al; Clin. Chim. Acta, 79, 93 (1977).
10)K.Sugawara and A.Kihara; Eisei Kensa, 27(1), 106 (1978).
11)H.Okabe et al ; Clin.Chim.Acta, 80, 87 (1977).
Table 1. Effect of Various Chemicals on Choline oxidase
[The enzyme dissolved in 10mM Tris-HCl buffer, pH 8.0 contg. 2mM EDTA and 1.0 % KCl (5U/mL) was incubated with each chemical at 25 ℃ for 1hr.]
-
Chemical Concn.(mM) Residual activity(%) None ー 100 Metal salt 2.0 MgCl2 87 CaCl2 92 Ba(OAc)2 89 FeCl3 87 CoCl2 89 MnCl2 91 ZnCl2 88 CdCl2 92 NiCl2 91 CuSO4 92 Pb(OAc)2 87 AgNO3 80 HgCl2 48 2-Mercaptoethanol 2.0 90 PCMB 1.0 13 -
Chemical Concn.(mM) Residual activity(%) MIA 2.0 87 NEM 2.0 100 IAA 2.0 95 Hydroxylamine 2.0 77 EDTA 5.0 92 o-Phenanthroline 2.0 90 α,α′-Dipyridyl 1.0 91 Borate 50 94 NaF 2.0 92 NaN3 2.0 92 Triton X-100 0.10 % 96 Brij 35 0.10 % 92 Tween 20 0.10 % 95 Span 20 0.10 % 94 Na-cholate 0.10 % 96 SDS 0.05 % 95 DAC 0.05 % 91
-
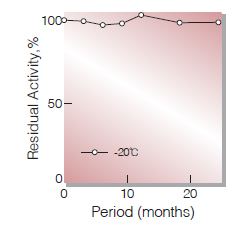
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
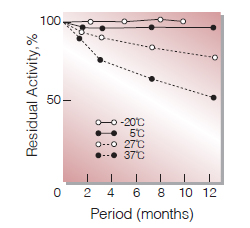
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
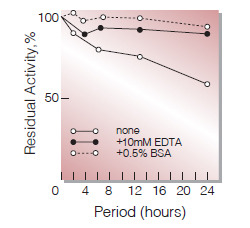
Fig.3. Stability (Liquid form at 37 ℃)
enzyme concentration: 1.0mg/mL buffer composition: 0.1M K-phosphate buffer, pH7.5
-
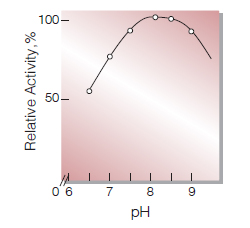
Fig.4. pH-Activity
(37 ℃, in 50mM K-phosphate buffer)
-
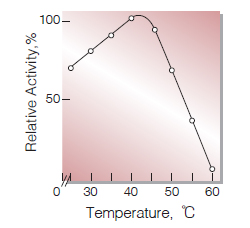
Fig.5. Temperature activity
(in 50mM K-phosphate buffer,pH7.5)
-
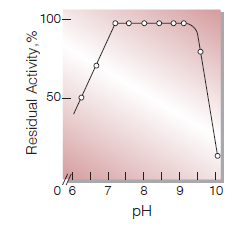
Fig.6. pH-Stability
30 ℃, 2hr-treatment with 50mM buffer solution:pH6.0-9.0, K-phosphate; pH9.0-10.0, glycine-NaCl-NaOH.
-
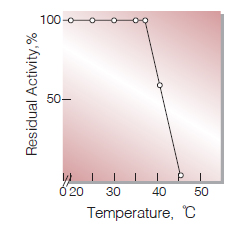
Fig.7. Thermal stability
15min-treatment with 50mM K-phosphate buffer, pH7.5
活性測定法(Japanese)
1. 原理

4-AminoantipyrineとPhenolの酸化縮合生成物であるQuinoneimine色素を500nmで測定し,上記反応で生成したH2O2量を定量する。
2.定義
下記条件下で1分間に1マイクロモルのH2O2を生成する酵素量を1単位 (U)とする。
3.試薬
- 2.1 %塩化コリン溶液(2.1gの塩化コリンを0.1MTris-HCl緩衝液,pH8.0で溶解し100 mLとする)
- 1.0 % 4-AA水溶液(1.0gの4-アミノアンチピリンを蒸留水に溶解して100 mLとする)(褐色瓶中で4 ℃保存)
- 1.0 %フェノール水溶液(1.0gのフェノールを蒸留水に溶解して100 mLとする)(褐色瓶中で4 ℃保存)
- 0.1M Tris-HCl緩衝液,pH8.0〔12.1gのトリス(MW=121.14)を約800 mLの蒸留水で溶解し,2.0NHClでpH8.0(25 ℃)に調製した後1000 mLにする〕
酵素溶液:酵素標品を予め氷冷した0.1M Tris-HCl緩衝液,pH8.0で溶解し,2.0mM EDTAと1.0 %のKClを含む10mM Tris-HCl緩衝液,pH8.0で0.1〜0.5 U/mLに希釈する。
4.手順
1.下記反応混液を調製する(褐色瓶にて氷冷保存)。
| 97.0 mL | 基質溶液 | (A) |
|---|---|---|
| 1.0 mL | 4-AA水溶液液 | (B) |
| 2.0 mL | フェノール水溶液 | (C) |
| 5.0 mL | peroxidase(110プルプロガリン単位/mg) | |
2.反応混液3.0 mLをキュベット(d = 1.0 cm)にとり,37℃で約5分間予備加温する。
3.酵素溶液0.05 mLを添加し,ゆるやかに混和し,反応混液を対照に37℃に制御された分光光度計で500nmの吸光度変化を3〜4分間記録し,その初期直線部分から1分間あたりの吸光度変化を求める(ΔOD/min)。
5.計算式
U/mL =
-
ΔOD/min×3.05(mL)
12.0×1/2×1.0×0.05(mL)
×希釈倍率
| = ΔOD/min×10.17×希釈倍率 | |
| U/mg | =U/mL×1/C |
| 12.0 | : Quinoneimine色素の上記測定条件下でのミリモル分子吸光係数(cm2/micromole) |
| 1/2 | : 酸素反応で生成したH2O2の1分子のから形成するQuinoneimine色素は1/2分子である事による係数。 |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/mL) |
CONTACT
-
For inquiries and cosultations regarding our products, please contact us through this number.
- HEAD OFFICE+81-6-6348-3843
- Inquiry / Opinion
