- HOME
- Enzyme List
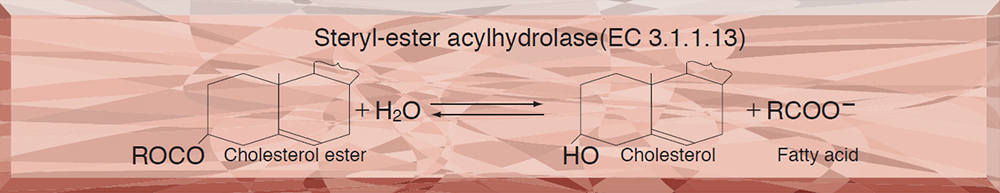
- COE-311 CHOLESTEROL ESTERASE
COE-311
CHOLESTEROL ESTERASE from Pseudomonas sp.
PREPARATION and SPECIFICATION
| Appearance | Light brown amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅢ 100 U/mg-solid or more (containing approx. 40 % of stabilizers) |
|
| Contaminants | Catalase | ≤1.0×10-2 % |
| Stabilizers | Mg2+, Na-cholate, BSA | |
PROPERTIES
| Stability | Stable at −20 ℃ for at least one year (Fig.1) |
|---|---|
| Molecular weight | approx. 300,000 |
| Isoelectric point | 5.9±0.1 |
| Michaelis constants | 5.4×10-5 M (Linoleate),6.6×10-5 M (Oleate), |
| 3.7×10-5 M (Linolenate),1.5×10-4 M (Palmitate) | |
| 1.2×10-4 M (Myristate),2.3×10-5 M (Stearate) | |
| Inhibitors | Hg2+, Ag+, ionic detergents |
| Optimum pH | 7.0−9.0(Fig.4) |
| Optimum temperature | 40 ℃(Fig.5) |
| pH Stability | pH 5.0−9.0 (25 ℃, 24 hr)(Fig.6) |
| Thermal stability | below 55 ℃ (pH 7.5, 10 min)(Fig.7) |
| Substrate specificity | (Table 1) |
| Effect of various chemicals | (Table 2) |
APPLICATIONS
This enzyme is useful for enzymatic determination of total cholesterol in combination with cholesterol oxidase (COO-311, COO-321, COO-331) in clinical analysis.
ASSAY
Principle
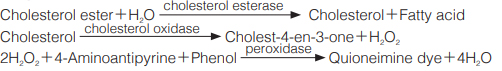
The formation of quinoneimine dye by reaction between 4-aminoantipyrine and phenol is measured at 500 nm by spectrophotometry.
Unit definition
One unit causes the formation of one micromole of hydrogen peroxide (half a micromole of quinoneimine dye) per minute under the conditions detailed below.
Method
Reagents
| A. 0.2M K-Phosphate buffer, pH 7.0 | |
|---|---|
| B. Cholesterol linoleate solution | To 39 mg of cholesterol linoleate, add 2.0 mL of isopropanol and dissolve completely by heating gently. Mix with about 80 mL of 1.0 % (v/v) hot Triton X-100 solution (preheated at 72−74 ℃) to the cholesterol linoleate solution and keep the solution in a hot water bath (72−74 ℃), stirring for 30 minutes. The solution will turn clear and then cloudy. Cool under running water with gentle agitation until the temperature of the solution falls to room temperature. Add 600mg of sodium cholate and dissove. Make up the solution to 100 mL with 1.0 % Triton X-100 solution. This solution is stable at 4 ℃ for at least 5 days. |
| C. 4-AA solution | 1.76 % (1.76 g 4-aminoantipyrine/100 mL of H2O; store at 4 ℃ in a brownish bottle) |
| D. Phenol solution | 6.0 % (6.0 g phenol/100 mL of H2O; store at 4 ℃ in a brownish bottle) |
| E. POD solution | Horseradish peroxidase (Toyobo, GradeⅢ) 7,500 purpurogallin units/50 mL of 0.1M K-phosphate buffer, pH 7.0 (150 PU/mL, freshly prepared) |
| F. COD solution | Streptomyces sp. cholesterol oxidase (Toyobo, GradeⅢ) 1,500 U/5.0 mL of ice-cold H2O(300 U/mL, freshly prepared) |
| G. Enzyme diluent | 20 mM Potassium phosphate buffer, pH 7.5 containing 2 mM MgCl2, 0.5 mM EDTA-Na3 and 0.2 % BSA |
Procedure
1.Prepare the following working solution (for 50 tests) in a brownish bottle.
| 75 mL | Buffer solution | (A) |
|---|---|---|
| 50 mL | Substrate solution | (B) |
| 2.5 mL | 4-AA solution | (C) |
| 5.0 mL | Phenol solution | (D) |
| 5.0 mL | POD solution | (E) |
| (This solution is stable at 4℃ for at least 5 days.) | ||
| Concentration in assay mixture | |
|---|---|
| Potassium phosphate buffer | 0.11 M |
| Cholesterol linoleate | 0.20 mM |
| 4-Aminoantipyrine | 1.5 mM |
| Phenol | 22 mM |
| EDTA | 17 μM |
| Isopropanol | 0.68 % |
| COD | approx. 10 U/mL |
| POD | approx. 5.1 U/mL |
2.Pipette 2.75 mL of working solution into a cuvette (d = 1.0cm) and equilibrate at 37 ℃ for approximately 5 minutes. Add 0.1 mL of COD solution (F), mix and keep at 37 ℃ for another 2 minutes.
3.Add 0.1 mL of the enzyme solution* and mix by gentle inversion.
4.Record the increase in optical density at 500 nm against water for 3 to 4 minutes in a spectrophotometer thermostated at 37 ℃, and calculate the ΔOD per minute from the initial linear portion of the curve (ΔODtest).
At the same time, measure the blank rate (ΔOD blank) using the same method as the test except that the enzyme diluent (G) is added instead of the enzyme solution.
*Dissolve the enzyme preparation in ice-cold enzyme diluent (G), and dilute to 0.08−0.22 U/mL with the same buffer, immediately before the assay.
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/mL) =
-
ΔOD/min (ΔOD test−ΔOD blank)×Vt×df
13.78×1/2×1.0×Vs
= ΔOD/min×4.282×df
Weight activity (U/mg) = (U/mL)×1/C
| Vt | : Total volume (2.95 mL) |
| Vs | : Sample volume (0.1 mL) |
| 13.78 | : Millimolar extinction coefficient of quinoneimine dye under the assay conditions (cm2/micromole) |
| 1/2 | : Factor based on the fact that one mole of H2O2 produces half a mole of quinoneimine dye. |
| 1.0 | : Light path length (cm) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/mL) |
REFERENCES
1)C.C.Allain, L.S.Poon, C.S.G.Chan, W.Richmond, and P.C.Fu; Clin.Chem., 20, 470 (1974).
2)Y.Kameno, N.Nakano, and S.Baba; Jap.J.Clin.Path., 24, 650 (1976).
Table 1. Substrate Specificity of Cholesterol esterase
[The reaction was carried out at 37℃ in 0.1M K-phoshate buffer, pH 7.0 contg. 0.2mM cholesterol ester, 0.33 % Triton X-100 and 0.2 % Na-cholate.]
-
Cholesterol ester Relative activity(%) Linoleate(18 :2) 100 Acetate(2 :0) 2.9 Propionate(3 :0) 21.3 Crotonate(4 :1) 0.0 Valerate(5 :0) 8.0 Caproate(6 :0) 17.3 Heptanoate(7 :0) 18.6 Caprylate(8 :0) 58.6 Nonanoate(9 :0) 48.0 Decylate(10 :0) 40.0 10-Undecenoate(11 :1) 114.6 Laurate(12 :0) 54.6 -
Cholesterol ester Relative activity(%) Tridecanoate(13 :0) 53.3 Myristate(14 :0) 49.3 Pentadecanote(15 :0) 46.6 Palmitate(16 :0) 33.5 Heptadecanoate(17 :0) 0.0 Stearate(18 :0) 8.0 Oleate(18 :1) 105.3 Lelaidate(18 :3) 166.5 Phenyl acetate 0.0 Cinnamate 0.0 Benzoate 58.6
Table 2. Effect of Various Chemicals on Cholesterol esterase
[The enzyme (13.5U/mL) was incubated at 25℃ for 1hr with each chemical.]
-
Chemical Concn.(mM) Residual activity(%) None ー 100 Metal salt 2.0 CaCl2 96 Ba(OAc)2 100 FeCl3 87 CoCl2 97 MnCl2 88 Zn(OAc)2 83 NiCl2 99 Pb(OAc)2 95 AgNO3 51 HgCl2 14 NEM 2.0 100 PCMB 2.0 100 -
Chemical Concn.(mM) Residual activity(%) MIA 2.0 96 NaF 20.0 99 NaN3 20.0 100 EDTA 5.0 100 o-Phenanthroline 2.0 100 α,α′-Dipyridyl 2.0 99 Borate 20.0 100 Triton X-100 1.0 % 97 Brij 35 1.0 % 100 SDS 0.1% 8 Na-cholate 1.0 % 100 Taurocholate 0.1 % 100
EDTA, Ethylenediaminetetraacetate; SDS, Sodium dodecyl sulfate.
-
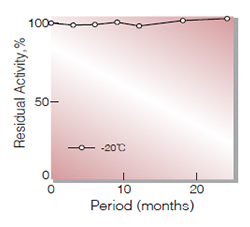
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
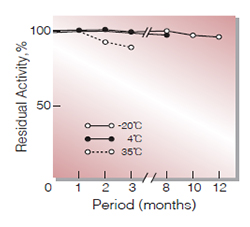
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
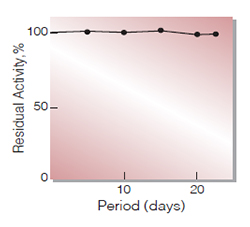
Fig.3. Stability (Liquid form at 5℃)
enzyme concentration:673.5U/mL buffer composition :0.1M K-phosphate buffer,pH7.0
-
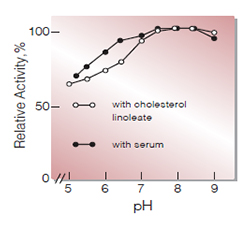
Fig.4. pH-Activity
(37℃,in 0.2 M K-phosphate buffer)
-
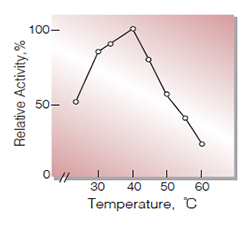
Fig.5. Temperature activity
(in 0.1M K-phosphate buffer,pH7.0)
-
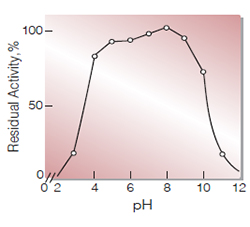
Fig.6. pH-Stability
25℃, 24hr-treatment with 0.2M Britton-Robinson buffer
-
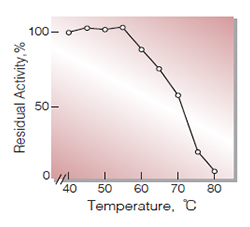
Fig.7. Thermal stability
10min-treatment with 20mM K-phosphate buffer,pH7.5 contg. 2mM MgCl2& 0.5mM EDTA・Na3
活性測定法(Japanese)
1. 原理

4-AAとフェノールの酸化縮合生成物であるQuinoneimine色素を500nmで測定し,上記反応で生成したH2O2量(加水分解されたCholesterol esterの量)を定量する。
2.定義
下記条件下で1分間に1マイクロモルのcholesterol esterを加水分解する酵素量を1単位 (U)とする。
3.試薬
- 0.2M K-リン酸緩衝液, pH7.0
- コレステロールリノレート溶液〔39mgのコレステロールリノレートを精秤し,2.0 mLのイソプロパノールを加え完全に加温溶解する。それを予め72〜74℃に加温しておいた約80 mLの1.0 %(V/V)トリトンX-100溶液と混和し,更に72〜74℃の温湯中で攪拌しながら30分間保ったのち,流水中で室温まで冷却する。次いで600mgのコール酸ナトリウムを加えて溶解させ,前記1.0 %トリトンX-100溶液で最終液量を100 mLとする〕(4℃保存で5日間は使用可能)
- 1.76 %4-AA水溶液〔1.76gの4-アミノアンチピリンを100 mLの蒸留水に溶解し,褐色瓶中で4℃に保存する〕
- 6.0 %フェノール水溶液〔6.0gのフェノールを100 mLの蒸留水に溶解し,褐色瓶中で4℃に保存する〕
- POD溶液〔西洋ワサビ由来ペルオキシダーゼ(東洋紡製,GradeⅢ)7,500プルプロガリン単位を50 mLの0.1M K-リン酸緩衝液, pH7.0に溶解する(150PU/mL)〕(用時調製)
- COD溶液〔ストレプトミセス属由来のコレステロールオキシダーゼ(東洋紡製,GradeⅢ)1,500単位を5.0 mLの氷冷蒸留水に溶解する(300 U/mL)〕(用時調製)
酵素溶液:酵素標品を予め氷冷した2mM MgCl20.5mM ETDA-Na3及び0.2 % BSAを含む20mM K-リン酸緩衝液, pH7.5で溶解し,分析直前に同緩衝液で0.08〜0.22U/ mLに希釈する。
4.手順
1.下記反応混液を(50テスト)を褐色瓶中で調製する(4℃保存で5日間は使用可能)。
| 75 mL | K-リン酸緩衝液 | (A) |
| 50 mL | 基質溶液 | (B) |
| 2.5 mL | 4-AA水溶液 | (C) |
| 5.0 mL | フェノール水溶液 | (D) |
| 5.0 mL | POD溶液 | (E) |
2.反応混液2.75 mLを試験管に採り,37℃で約5分間予備加温し,0.1 mLのCOD溶液を加えて更に2分間加温する。
3.酵素溶液0.1 mLを添加し,ゆるやかに混和後,水を対照に37℃に制御された分光光度計で500nmの吸光度変化を3〜4分間記録し,その初期直線部分から1分間あたりの吸光度変化を求める(ΔOD test)。
4.盲検はCOD添加液2に酵素溶液の代りに酵素希釈液(2mM MgCl2, 0.5mM EDTA-Na3及び0.2 % BSA)を含む20mMK-リン酸緩衝液, pH7.5を0.1 mL加え,上記同様に操作を行って1分間当りの吸光度変化を求める(ΔODblank)。
5.計算式
U/mL =
-
ΔOD/min (ΔOD test−ΔOD blank)×2.95(mL)×希釈倍率
13.78×1/2×1.0×0.1(mL)
| = ΔOD/min×4.282×希釈倍率 | |
| U/mg | =U/mL×1/C |
| 13.78 | : Quinoneimine色素の上記測定条件下でのミリモル分子吸光係数 (cm2/micromole) |
| 1/2 | : H2O2の1分子のから形成するQuinoneimine色素は1/2分子である事による係数 |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/mL) |
CONTACT
-
For inquiries and cosultations regarding our products, please contact us through this number.
- HEAD OFFICE+81-6-6348-3843
- Inquiry / Opinion
