- HOME
- Enzyme List
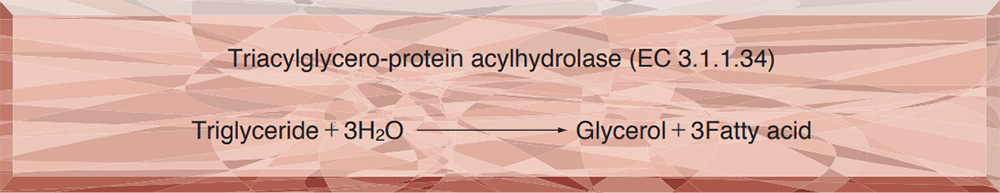
- LPL-311 LIPOPROTEIN LIPASE
LPL-311
LIPOPROTEIN LIPASE from Pseudomonas sp.
PREPARATION and SPECIFICATION
| Appearance | Light brown amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅢ 20 U/mg-solid or more (containing approx. 80 % of stabilizers) |
|
| Contaminants | Phosphatase | ≤ 1.0×10-3 % |
| Catalase | ≤ 2.0×10-2 % | |
| NADH oxidase | ≤ 1.0×10-3 % | |
| Cholesterol oxidase | ≤ 2.0×10-3 % | |
| Stabilizers | Mg2+, Na-cholate, BSA | |
PROPERTIES
| Stability | Stable at −20 ℃ for at least one year(Fig.1) |
|---|---|
| Molecular weight | approx. 134,000 |
| Isoelectric point | 5.95±0.05 |
| Inhibitors | Hg2+, Ag+, ionic detergents |
| Optimum pH | 7.0−9.0(Fig.4) |
| Optimum temperature | 45−50 ℃(Fig.5) |
| pH Stability | pH 7.0−9.0 (25 ℃, 20 hr)(Fig.6) |
| Thermal stability | below 55 ℃ (pH 7.0, 10 min)(Fig.7) |
| Substrate specificity | (Table 1) |
| Effect of various chemicals | (Table 2) |
APPLICATIONS
This enzyme is useful for enzymatic determination of triglyceride in serum when in combination L-αglycerophosphate oxidase (G3O-321) andglycerol kinase (GYK-301,GYK-311). Usually, the reaction can be completed in 5 minutes at 37 ℃ by using 2.5〜3.0 units of the enzyme per test (3.0 mL) at a pH of around 7.0.
ASSAY
Principle

The formation of quinoneimine dye is measured at 545 nm by spectrophotometry.
Unit definition
One unit causes the formation of one micromole of glycerol (half a micromole of quinoneimine dye) per minute under the conditions detailed below.
Method
Reagents
| A. Olive oil emulsion | Sonicate the mixture of 5.0 g of olive oil[reagent grade (highly refined, low acidity)]and 5.0 mL of 5.0 % Triton X-100 solution (B) for 10 minutes (20 KHz). To the oil emulsion, add 25 mL of 4.0 % BSA solution (C) and 15 mL of 0.1 M potassium phosphate buffer, pH 7.0 (D), and mix. (should be freshly prepared) | |
|---|---|---|
| B. Triton X-100 solution | 5.0 % (5.0 mL Triton X-100/100 mL of H2O) | |
| C. BSA solution | 4.0 %[4.0 g bovine serum albumin/100 mL of H2O] | |
| D. K-phosphate buffer, pH 7.0 | 0.1 M | |
| E. TCA solution | 0.2 M (33 g trichloroacetic acid/1,000 mL of H2O) | |
| F. MES-NaOH buffer | 50 mM MES buffer, pH 6.5: Dissolve 9.76 g of 2-(N-morpholino)-ethanesulfonic acid (MW = 195.23) in approx. 850 mL of H2O, and, after adjusting the pH to 6.5 with 5.0 N NaOH, make up to 1,000 mL with H2O. | |
| G. Color developing reagent | Dissolve the following chemicals and enzymes in 200 mL of 50 mM MES buffer (F) in the following order: | |
| 4.0 mL | Triton X-100 solution (B) | |
| 0.04 mL | N,N-diethyl-m-toluidine (Stir until completely dissolved) | |
| 4.0 mg | 4-Aminoantipyrine | |
| 24.2 mg | ATP・Na2・3H2O | |
| 40.7 mg | MgCl2・6H2O | |
| 200 units | Glycerol kinase (Toyobo, grade III) | |
| 500 units | L-α-glycerophosphate oxidase (Toyobo, grade III) | |
| 300 units | Peroxidase (purpurogallin units; Toyobo, grade III) | |
| (Stable for one week if stored at 4 ℃ in a brownish bottle) | ||
| H. Enzyme diluent | 20 mM Potassium phosphate buffer, pH 7.5, containing 2.0 mM MgCl2 and 0.5 mM EDTA-Na3 | |
Procedure
(1st step)
1. Pipette 2.0 mL of olive oil emulsion (A) into a test tube and equilibrate at 37 ℃ for approximately 5 minutes.
2. Add 0.2 mLl of the enzyme solution* and mix.
3. After exactly 15 minutes at 37 ℃, add 2.0 mL of TCA solution (E) to stop the reaction and remove the precipitate by filtration with filter paper.
| Concentration in assay mixture | |
|---|---|
| Potassium phosphate buffer | 29.1 mM |
| Olive oil | 90.9 mg/mL |
| MgCl2 | 0.18 mM |
| Triton X-100 | 9.1 % |
| EDTA | 45 μM |
| BSA | 1.8 % |
(2nd step)
4.Pipette 0.05 mL of the filtrate thus obtained into a test tube.
5.Add 3.0 mL of color developing reagent (G) and incubate at 37 ℃ for 15 minutes.
6.Measure the optical density at 545 nm against water (OD test).
At the same time, prepare the blank by mixing 2.0 mL of olive oil emulsion (A), after incubation for 10 minutes at 37 ℃, with 2.0 mL of TCA solution, followed by addition of the enzyme solution (1st step). Using the filtrate obtained from the mixture, carry out the 2nd step, using the same procedure as in the test, and measure the optical density at 545 nm (OD blank).
*Dissolve the enzyme preparation in ice-cold enzyme diluent (H) and dilute to 0.4−1.2 U/mL with the same buffer, immediately before assay.
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/mL) =
-
ΔOD (OD test−OD blank)×Vt-1×Vt-2×df
28.2×1/2×1.0×t×Vs-1×Vs-2
= ΔOD×6.057×df
Weight activity (U/mg) = (U/mL)×1/C
| Vt-1 | : Total volume in 1st step (4.2 mL) |
| Vt-2 | : Total volume in 2nd step (3.05 mL) |
| Vs-1 | : Sample volume in 1st step (0.2 mL) |
| Vs-2 | : Sample volume in 2nd step (0.05 mL) |
| 28.2 | : Millimolar extinction coefficient of quinoneimine dye under the assay condition (cm2/micromole) |
| 1/2 | : Factor based on the fact that one mole of H2O2 produces half a mole of quinoneimine dye |
| 1.0 | : Light path length (cm) |
| t | : Reaction time in 1st step (15 minutes) |
| df | : Dilution factor |
| C | : Enzyme concentration in dissolution (c mg/mL) |
REFERENCES
1) T.Saiki, Y.Takagi, T.Suzuki, T.Narasaki, G.Tamura and K.Arima; Agric. Biol. Chem. (Tokyo), 33, 414 (1969).
2) T.Yamaguchi, N.Muroya, M.Isobe and M.Sugiura; Agric. Biol. Chem. (Tokyo), 37, 999 (1973).
Table 1. Substrate Specificity of Lipoprotein lipase (Substrate :10 %)
-
Substrate Relative
activity(%)Olive oil 94 Triolein (18 :1) 100 Tripalmitin (16 :0) 2 Trimyristin (14 :0) 7 Trilaurin (12 :0) 4 Tricaprin (10 :0) 17 -
Substrate Relative
activity(%)Tricaprylin (8 :0) 64 Tricaproin (6 :0) 2 Tributyrin (4 :0) 2 Tripropionin (3 :0) 2 Triacetin (2 :0) 1
Number of carbon atoms to number of double bonds is given in parenthesis.
Table 2. Effect of Various Chemicals on Lipoprotein lipase
[The enzyme (2.5 U/mL) was incubated at 25 ℃ for 1 hr with each chemical.]
-
Chemical Concn.(mM) Residual
activity(%)None - 100 CaCl2 2.0 95 Ba(OAc)2 2.0 92 FeCl3 2.0 80 CoCl2 2.0 90 MnCl2 2.0 85 Zn(OAc)2 2.0 86 NiCl2 2.0 97 Pb(OAc)2 2.0 80 AgNO3 2.0 47 HgCl2 2.0 5 CdCl2 1.0 82 CrCl2 1.0 42 SnCl2 1.0 49 CuSO4 1.0 58 NEM 2.0 98 -
Chemical Concn.(mM) Residual
activity(%)PCMB 2.0 100 MIA 2.0 98 NaF 20.0 95 NaN3 20.0 97 EDTA 5.0 100 o-Phenanthroline 2.0 100 α,α′-Dipyridyl 2.0 94 Borate 20.0 100 Triton X-100 1.0 % 100 Brij 35 1.0 % 100 SDS 0.1 % 4 Tween 20 0.1 % 89 Span 20 0.1 % 100 Na-cholate 1.0 % 93 Taurocholate 0.1 % 100
Ac, CH3CO; NEM, N-Ethylmaleimide; PCMB, p-Chloromercuribenzoate; MIA, Monoiodoacetate; EDTA, Ethylenediamineteraacetate; SDS, Sodium dodecyl sulfate.
-
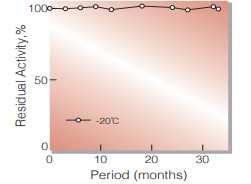
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
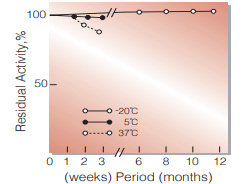
Fig.2. Stability (Powder form)
(kept under dry conditions)
-
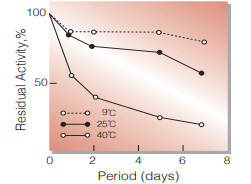
Fig.3. Stability (Liquid form)
in 20 mM K-phosphate buffer,pH 7.5 (contg. 2.0 mM MgCl2, 0.5 mM EDTA.Na3, 0.005 % NaN3) enzyme concn.:4 U/mL
-
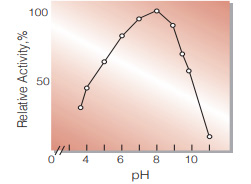
Fig.4. pH-Activity
(37 ℃,in 0.1 M Britton-Robinson buffer)
-
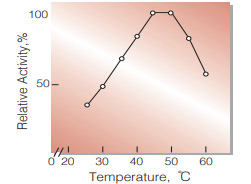
Fig.5. Temperature activity
(in 0.1 M K-phosphate buffer, pH 7.0)
-
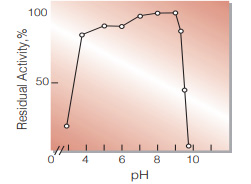
Fig.6. pH-Stability
25 ℃,20 hr-treatment with 0.1 M Britton-Robinson buffer
-
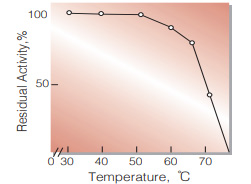
Fig.7. Thermal stability
10 min-treatment with 0.1 MK-phosphate buffer, pH 7.0
活性測定法(Japanese)
1. 原理

4-AminoantipyrineとN,N-Diethyl-m-touluidineの酸化縮合生成物であるQuinoneimine色素を545nmで測定し,上記反応で生成したH2O2を定量する。
2.定義
下記条件で1分間に1マイクロモルのGlycerol(1/2マイクロモルのQuinoneimine色素)を生成する酵素量を1単位(U)とする。
3.試薬
- オリーブ油エマルジョン液〔オリーブ油 (ナカライテスク製,リパーゼ測定用特製試薬)5.0gと5.0 %トリトンX-100溶液(B)5.0 mLの混合液を10分間超音波処理し (20KHz),エマルジョンを調製する。次いでこのエマルジョンに4.0 % BSA水溶液(C)25 mLと0.1MK-リン酸緩衝液,pH7.0 (D)15 mLを添加混合する〕(用時調製)
- 5.0 %(V/V)トリトンX-100溶液 (5.0 mLのTriton X100を100 mLの蒸留水に溶解する)
- 4.0 %牛血清アルブミン(BSA)水溶液〔4.0gの牛血清アルブミンを100 mLの蒸留水に溶解する〕
- 0.1MK-リン酸緩衝液,pH7.0
- 0.2Mトリクロル酢酸(TCA)溶液(33gのトリクロル 酢酸を1,000 mLの蒸留水に溶解する)
- 50mM MES緩衝液,pH6.5〔9.76gの2-(Nmorpholino) ethanesulfonic acid (MW= 195.23)を約850 mLの蒸留水に溶解し,pHを5.0N NaOHで6.5に調整後,蒸留水で1,000 mLとする〕
-
発色試薬〔200 mLの50m M M E S緩衝液, pH6.5(F)に下記順序で試薬及び酵素を溶解する〕
4.0 mL 5.0 %トリトンX-100溶液 (B) 0.04 mL N,N-Diethyl-m-toluidine(完全に溶解するまで攪拌する) 4.0 mg 4-アミノアンチピリン 24.2 mg ATP・Na2・3H2O 40.7 mg MgCl2・6H2O 200 単位 Glycerol kinase(東洋紡製, GradeⅢ) 500 単位 L-α-Glycerophosphate oxidase(東洋紡製, GradeⅢ) 300 単位 Peroxidase (プルプロガリン単位)(東洋紡製, GradeⅢ)
(上記発色試薬は4℃,褐色瓶中で保存すれば1週間は使用可能)
酵素溶液:酵素標品を予め氷冷した2.0mM MgCl2及び0.5mM EDTA-Na3を含む20mMK-リン酸緩衝液,pH7.5で溶解し,分析直前に同緩衝液で0.4〜1.2U/mLに希釈する。
4.手順
1.オリーブ油エマルジョン液(A)2.0 mLを試験管に採り,37℃で約5分間予備加温する。
2.酵素溶液0.2 mLを加え,反応を開始する。
3.37℃で正確に15分間反応させた後,TCA溶液(E)2.0 mLを加えて反応を停止する。
4.生成する不溶物を濾紙濾過で除く。
5.濾液の0.05 mLを試験管に採り,発色試薬(G)3.0 mLを加えて混合した後,37℃にて15分間加温し,545nmにおける吸光度を測定する(OD test)。
6.盲検はオリーブ油エマルジョン液(A)2.0 mLを37℃で15分間放置後,TCA溶液(E)2.0 mLを加え,次いで酵素溶液0.2 mLを加えて調製し,以下上記同様 (④〜⑤)に操作して吸光度を測定する(OD blank)。
5.計算式
U/mL =
-
ΔOD (OD test−OD blank)×4.2(mL)×3.05(mL)×希釈倍率
28.2×1/2×1.0×15(分)×0.2(mL)×0.05(mL)
| = ΔOD×6.057×希釈倍率 | |
| U/mg | = U/mL×1/C |
| 28.2 | : Quinoneimine色素の上記測定条件下でのミリモル分子吸光度係数(cm2/micromole) |
| 1/2 | : H2O2の1分子から形成するQuinoneimine色素は1/2分子である事による係数 |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/mL) |
CONTACT
-
For inquiries and cosultations regarding our products, please contact us through this number.
- HEAD OFFICE+81-6-6348-3843
- Inquiry / Opinion
