- HOME
- Enzyme List
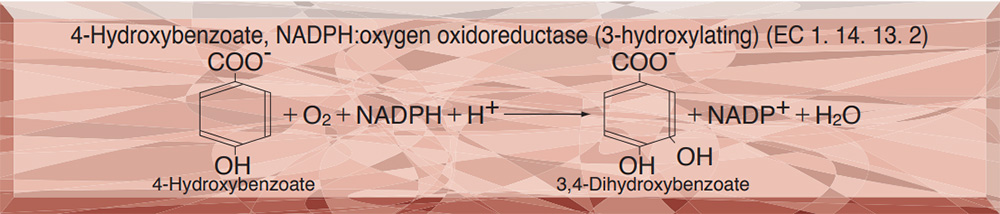
- HBH-311 p-HYDROXYBENZOATE HYDROXYLASE
-
HBH-311
p-HYDROXYBENZOATE HYDROXYLASE from Microorganism
PREPARATION and SPECIFICATION
| Appearance | Yellowish amorphous powder, lyophilized | |
|---|---|---|
| Activity | GradeⅢ 20 U/mg-solid or more (containing approx. 40 % of stabilizers) |
|
| Contaminant | NADPH oxidase ≤ 1.0×10-1 % | |
| Stabilizers | Sugars, FAD | |
PROPERTIES
| Stability | Stable at −20 ℃ for at least one year(Fig.1) |
|---|---|
| Molecular weight | 55,000〜60,000 |
| Michaelis constants | 2.0×10-5 M (p-Hydroxybenzoate), 4.0×10-5 M (NADPH) |
| Structure | One mol of FAD per mol of enzyme |
| Inhibitors | Ag+, Hg2+, PCMB, SDS |
| Optimum pH | 7.7−7.9(Fig.3) |
| Optimum temperature | 35 ℃(Fig.4) |
| pH Stability | pH 5.0−7.5 (25 ℃, 72 hr)(Fig.5) |
| Thermal stability | below 40 ℃ (pH 6.0, 15 min)(Fig.6) |
| Substrate specificity | (Table 1) |
| Effect of various chemicals | (Table 2) |
APPLICATIONS
This enzyme is useful for enzymatic determination of choline esterase in combination with protocatechuate 3, 4-dioxygenase (PCO-302)
ASSAY
Principle

The formation of NADPH is measured at 340 nm by spectrophotometry.
Unit definition
One unit causes the oxidation of one micromole of NADPH per minute under the conditions described below.
Method
Reagents
| A. Tris-malate buffer, pH 8.2 | 50 mM: Dissolve 3.03 g of Tris (MW = 121.14) in approx. 300 mL of H2O, and, after adjusting the pH to 8.2 at 25 ℃ with 1.0 M maleic acid, make up to 500 mL with H2O. | |
|---|---|---|
| B. p-hydroxybenzoate solution | 5.0 mM: 80 mg of sodium p-hydroxybenzoate / 100 mL of buffer solution A (should be freshly prepared). | |
| C. FAD solution | 0.2 mM: 19 mg of FAD・Na2 / 100 mL of buffer solution A (should be freshly prepared) | |
| D. NADPH solution | 3.0 mM: 272 mg NADPH・Na4・4H2O / 100 mL of buffer solution A (should be freshly prepared) | |
| E. Enzyme diluent | 50mM Potassium phosphate buffer, pH 6.0, containing 0.2 % BSA | |
Procedure
1. Prepare the following working solution (for 10 tests) in a brownish bottle and store on ice.
| 21.0 mL | Buffer solution | (A) |
| 3.0 mL | Substrate solution | (B) |
| 3.0 mL | FAD solution | (C) |
| 3.0 mL | NADPH solution | (D) |
| Concentration in assay mixture | |
|---|---|
| Tris-malate buffer | 49 mM |
| p-Hydroxybenzoate | 0.49 mM |
| FAD | 20 μM |
| NADPH | 0.30 mM |
2. Pipette 3.0 mL of working solution into a cuvette (d = 1.0 cm) and equilibrate at 37 ℃ for approximately 5 minutes.
3. Add 0.05 mL of the enzyme solution* and mix by gentle inversion.
4. Record the decrease in optical density at 340nm against water for 3 to 4 minutes with a spectrophotometer thermostated at 37 ℃ and calculate the ΔOD per minute from the portion of the curve from 1.5 to 3 minutes (ΔOD test).
At the same time, measure the blank rate (ΔOD blank) using the same method as the test except that the enzyme diluent (E) is added instead of enzyme solution.
*Dissolve the enzyme preparation in ice-cold enzyme diluent (E) (1.0 mg/mL or more) and dilute to 0.2−0.6 U/mL with the same buffer, immediately before the assay.
Calculation
Activity can be calculated by using the following formula :
Volume activity (U/mL) =
-
ΔOD/min (ΔOD test−ΔOD blank)×Vt×df
6.22×1.0×Vs
= ΔOD/min×9.8×df
Weight activity (U/mg) = (U/mL)×1/C
| Vt | : Total volume (3.05 mL) |
| Vs | : Sample volume (0.05 mL) |
| 6.22 | : Millimolar extinction coefficient of NADPH (cm2/micromole) |
| 1.0 | : Light path length (cm) |
| df | : Dilution factor |
| C | : Enzyme concentration (c mg/mL) |
REFERENCES
1) H.Shoun and K.Arima; Protein, Nucleic acid and Enzyme, 25, 820, (1980).
2) K.Yano and K.Arima; Agric.Biol.Chem., 33, 689 (1969).
3) K.Hosokawa and R.Y.Stanier; J.Biol.Chem., 241, 2453 (1966).
Table 1. Substrate Specificity of p-Hydroxybenzoate hydroxylase
-
Substrate(0.5mM) Relative activity(%) p-Hydroxybenzoic acid 100 Methyl-p-hydroxybenzoic acid <0.05 Ethyl-p-hydroxybenzoic acid <0.05 n-Propyl-p-hydroxybenzoic acid <0.05 m-Hydroxybenzoic acid <0.05 o-Hydroxybenzoic acid -
Substrate Relative activity(%) Protocatechuic acid 3.3 β-Resorcylic acid 4.5 Gentisic acid <0.05 p-Chlorobenzoic acid <0.05 p-Aminobenzoic acid 0.12
Table 2. Effect of Various Chemicals on p-Hydroxybenzoate hydroxylase (Residual activity after 1 hr-treatment at 30℃)
-
Chemical Concn.(mM) Residual
activity(%)None - 100 Metal salt 1.0 CoCl2 106 ZnCl2 94 CuSO4 103 AgNO3 0 MgSO4 107 BaCl2 107 FeCl3 106 MnCl2 90 NiCl2 104 CaCl2 97 SnCl2 102 HgCl2 1.1 CrCl2 93 CdCl2 104 FeSO4 90 -
Chemical Concn.(mM) Residual
activity(%)MIA 1.0 91 PCMB 1.0 3.7 NaN3 1.0 97 NaF 1.0 96 o-Phenanthroline 1.0 95 α,α′-Dipyridyl 1.0 90 EDTA 5.0 96 Borate 50 104 Tween 20 0.1 % 91 Brij 35 0.1 % 101 Span 20 0.1 % 94 Triton X-100 0.1 % 97 Na-cholate 0.1 % 88 SDS 0.05 % 34
MIA, Monoiodoacetate; PCMB, p-Chloromercuribenzoate; EDTA, Ethylenediaminetetraacetate; SDS, Sodium dodecyl sulfate.
-
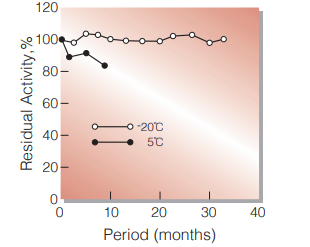
Fig.1. Stability (Powder form)
(kept under dry conditions)
-
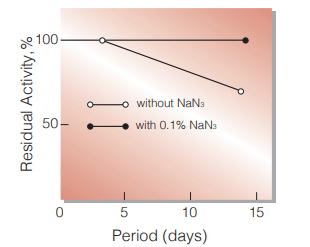
Fig.2. Stability (Liquid form at 25 ℃)
enzyme concentration:500 U/mL buffer compostion:50 mM K-phosphate buffer,p H6.0
-
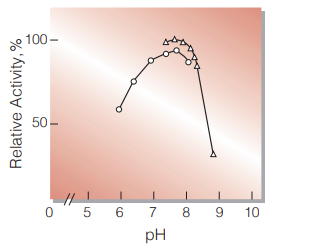
Fig.3. pH-Activity
37 ℃ in 50 mM buffer solution: ○̶̶○,K-phosphate; △̶̶△,Trismalate
-
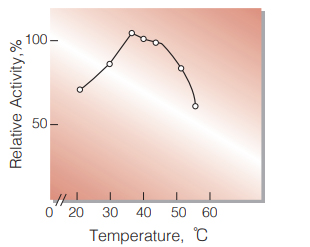
Fig.4. Temperature activity
(in 50 mM Tris-malate buffer, pH 8.2)
-
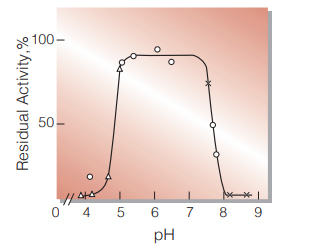
Fig.5. pH-Stability
25 ℃ in 72 hr-treatment with 50 mM buffer solution:△̶̶△,acetate; ○̶̶○,K-phosphate;×̶̶×,glycine-NaOH
-
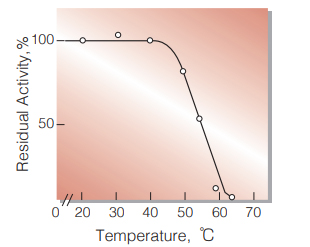
Fig.6. Thermal stability
15 min-treatment with 50 mM K-phosphate buffer,pH 6.0
活性測定法(Japanese)
1. 原理

NADPHの消失量を340nmの吸光度の変化で測定する。
2.定義
下記条件下で1分間に1マイクロモルのNADPHが酸化される酵素量を1単位(U)とする。
3.試薬
- 50mM Tris-malate緩衝液pH8.2(3.03gのTris(MW=121.14)を約300 mLの蒸留水で溶解し,1.0M マレイン酸でpHを8.2(25℃)に調整後,蒸留水で500 mLにする)
- 5.0mM p-ヒドロキシ安息香酸溶液(80mgのp-ヒドロキシ安息香酸ナトリウムを100 mLの緩衝液(A)で溶解する)(用時調製)
- 0.2mM FAD溶液(19mgのFAD・Na2を約100 mLの緩衝液(A)で溶解する)(用時調製)
- 3.0mM NADPH溶液(272mgのNADPH・Na4-4H2Oを約100 mLの緩衝液(A)で溶解する)(用時調製)
酵素溶液:酵素標品を予め氷冷した0.2 % BSAを含む50mM K-リン酸緩衝液pH6.0で溶解(1.0mg/mL以上)し,分析直前に同緩衝液で0.2〜0.6U/mLに希釈する。
4.手順
1.下記反応混液を調製する(用時調製し,褐色瓶で氷冷保存)。
| 21.0 mL | Tris-malate緩衝液 | (A) |
| 3.0 mL | 基質溶液 | (B) |
| 3.0 mL | FAD溶液 | (C) |
| 3.0 mL | NADPH溶液 | (D) |
2.反応混液3.0 mLをキュベット(d=1.0cm)に採り,37℃で約5分間予備加温する。
3.酵素溶液0.05 mLを添加し,ゆるやかに混和後,水を対照に37℃に制御された分光光度計で340nmの吸光度変化を1.5〜3.0分間記録し,その1.5〜3分間の吸光度から1分間当りの吸光度変化を求める(ΔODtest)。
4.盲検は反応混液に酵素溶液の代りに酵素希釈液(0.2% BSAを含む50mM K-リン酸緩衝液pH6.0)を0.05ml加え,上記同様に操作を行って,1分間当りの吸光度変化を求める(ΔOD blank)。
5.計算式
U/mL =
-
ΔOD/min (ΔOD test−ΔOD blank)×3.05(mL)×希釈倍率
6.22×1.0×0.05(mL)
| = ΔOD/min×9.8×希釈倍率 | |
| U/mg | = U/mL×1/C |
| 6.22 | : NADPHのミリモル分子吸光係数(cm2/micromole) |
| 1.0 | : 光路長(cm) |
| C | : 溶解時の酵素濃度(c mg/mL) |
CONTACT
-
For inquiries and cosultations regarding our products, please contact us through this number.
- HEAD OFFICE+81-6-6348-3843
- Inquiry / Opinion
